Broadcast
eve Conference Detail Block
Conference background
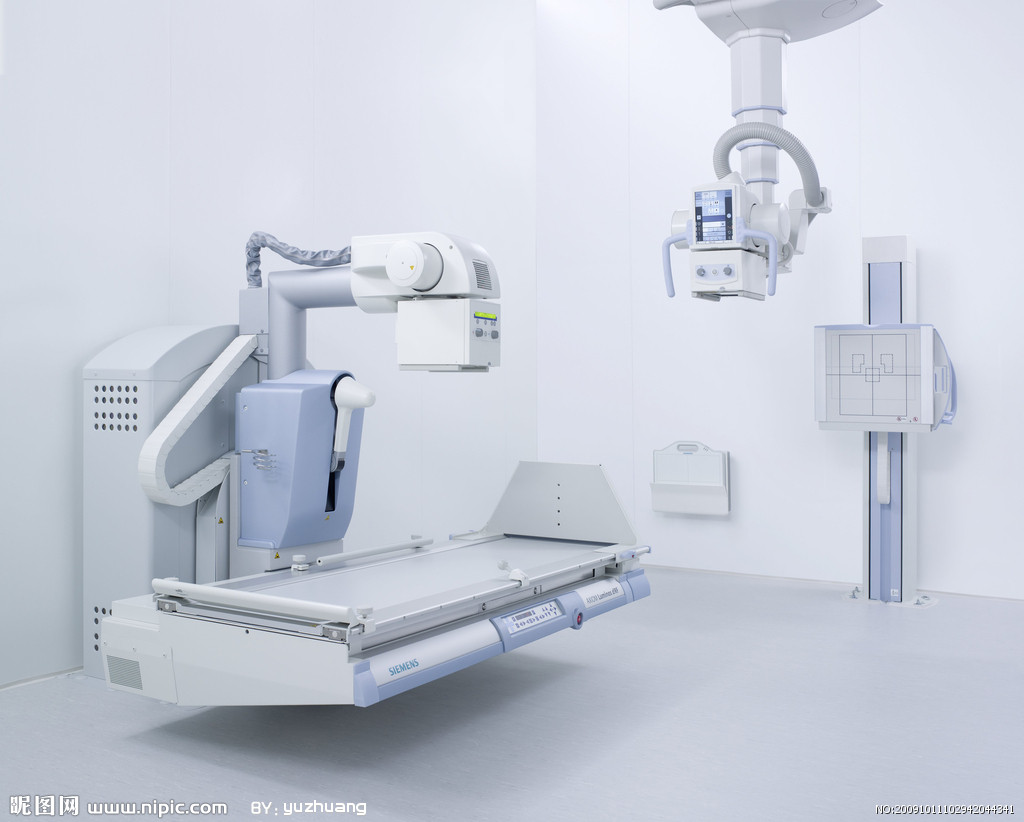
Medical plastics is a pearl in the pagoda in the plastics industry, have a vast market for its high-tech, high value-added, high growth speed, and have attracted much attention. As one of the important basic materials of modern society, application of plastic tentacles in the medical field also extends very widely, it has become a commonly used materials of medical device manufacturing, its application is incredibly. According to GIA' s earlier survey, total output of medical plastic in 2015 will exceed 10 billion pounds (about 4.5 billion kg), biocompatible, able to withstand the high heat disinfection, chemical resistance, high transparency and can show different shapes of plastic, is expected to be very popular in the pharmaceutical industry. "6th Plastics Technology in Medical Products Conference" held by Ringier, based on the previous five successful experience, with hot topic about the market trends, regulatory approvals, R & D and innovation, advanced processing technology and applications of large medical device manufacturers, brings together the national review agencies, clinicians, medical device manufacturers, third-party testing and certification bodies and other industry elite, depth discussion of market trends and application directions of medical plastics and medical equipment.Air Jordan 30.5 Shoes
Who will attend
By Industry:
Hospitals and medical institutions
Medical device manufacturers and suppliers
Medical plastic products manufacturers and suppliers
Molding equipment manufacturers and suppliers of medical plastics
Medical plastics raw materials manufacturers and suppliers
Third-party testing, certification bodies
Industry associations, research institutions, universities and research ins
Why you need to attend
1. Understand the latest regulations and global medical device market hotspots.
2. Discussion on the focus of the global medical device research and future trends.
3. Provide Omni-directional communication and consultation opportunities for participating companies to communicate with doctors and specialists.
4. Quality control, R&D ideas and experience sharing from reputed r leading companies.
5.